 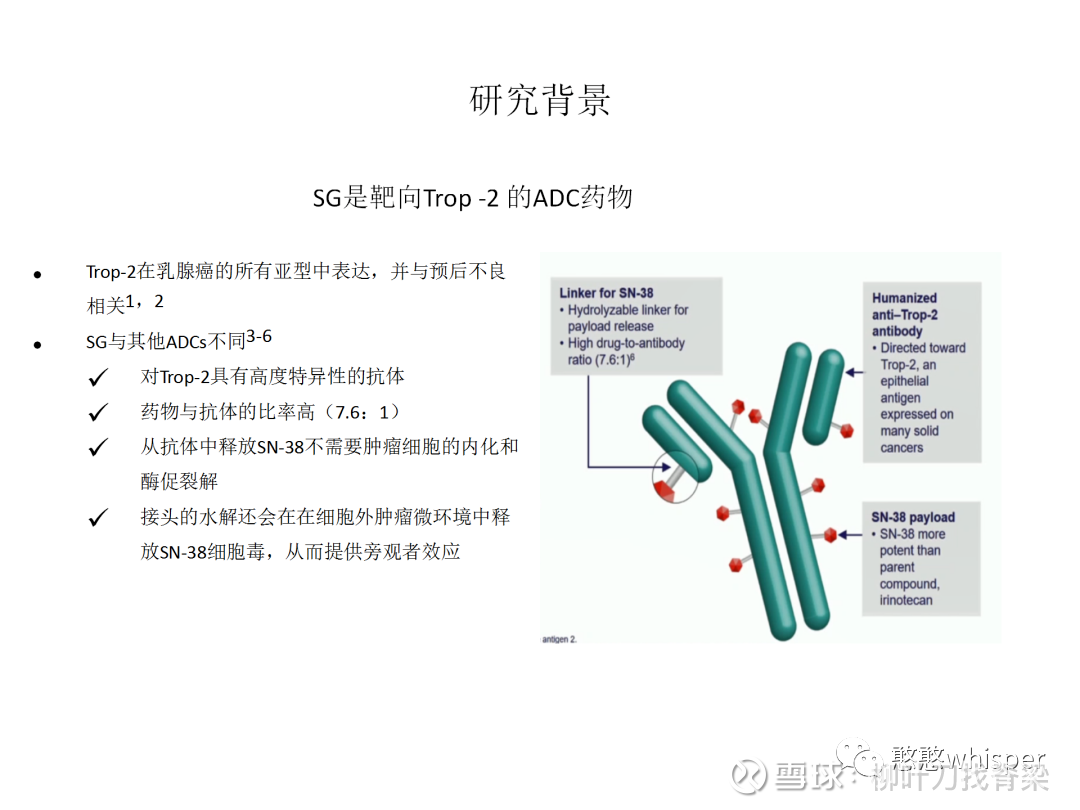
The character's first name was created by Adam Bray for the 2019 reference book Ultimate Star Wars, New Edition, who named him after Del Rey editor Erich Schoeneweiss. The character is also identified by name in the subtitles on the film's home video release. DS-1062 is a potential new medicine that has not been approved for any indication in any country. The character was not identified in the film, but was named in Star Wars: The Force Awakens: The Visual Dictionary, which was written by Pablo Hidalgo and released the same day as the film. Designed using Daiichi Sankyo’s proprietary DXd ADC technology, DS-1062 is comprised of a humanised anti-TROP2 monoclonal antibody attached to a topoisomerase I inhibitor payload by a tetrapeptide-based linker. Datoo was portrayed by Rocky Marshall in Star Wars: Episode VII The Force Awakens, which was released in 2015. He wore a teal First Order Army uniform with a crested command cap and a band on the left arm written in Aurebesh that commemorated the Imperial Warlord Kaplan. Personality and traits ĭatoo was a methodical officer who believed that the destructive power of Starkiller Base should command respect from all members of the First Order. 
Starkiller Base then exploded, killing Datoo along with it. As it became clear the base was going to be destroyed, Datoo was shocked to see Lieutenant Rodinon trying to leave the control room, and questioned where he was going, although the latter talked back and bluntly pointed out that they needed to evacuate or else they won't survive, citing that even the base's commander, Hux, had already evacuated. The weapon then began preparing to fire on the Ileenium system, where the Resistance headquarters was located however, Resistance forces then attacked the superweapon. Under orders from Supreme Leader Snoke, he oversaw the weapon as it destroyed the Hosnian system, the location of the New Republic Galactic Senate. In 34 ABY, Datoo was in charge of the primary fire control room on the First Order's Starkiller Base superweapon. Datoo was a human male military officer who held the rank of colonel in the First Order. Dato-DXd is an ADC, composed of a recombinant humanized anti-TROP2 IgG1 mAb conjugated with a Topo I inhibitor (DXd) via a tetrapeptide-based linker to reduced cysteine residues at the interchain disulfide bonds of datopotamab (Fig. MedwireNews is an independent medical news service provided by Springer Healthcare Ltd." Just look! We won't survive. The investigator concluded that “further study in breast cancer is needed,” adding that “the hormone receptor-positive cohort is now enrolling.” Thus, the majority of patients had a decrease in tumor volume and the majority of these responses occurred within the first 2 months of treatment, Bardia commented. Just one patient experienced disease progression, giving a disease control rate of 95%. These included a confirmed complete or partial response in 24% of patients and responses pending confirmation in 19%.  
Turning to efficacy results, the presenter said Dato-DXd had “impressive antitumor activity,” citing an objective response rate of 43% for the 21 patients assessed by blinded independent central review. The majority of treatment-emergent AEs were nonhematologic grade 1 or 2 side effects the most common any-grade events were stomatitis (63%), nausea (63%), fatigue (42%), vomiting (42%), alopecia (25%), cough (21%), and pruritus (21%), while anemia, headache, and constipation were each experienced by 17% of patients.īardia highlighted that there were no reports of drug-related interstitial lung disease, or grade 3 or more severe episodes of diarrhea or neutropenia. There were no serious or fatal TRAEs and none of the patients discontinued treatment because of AEs, but 25% of patients required a dose reduction, most commonly for stomatitis (13%) and mucosal inflammation (8%). These included taxanes (83%), platinum-based chemotherapy (50%), and immunotherapy (33%), as well as sacituzumab govitecan (8%) or a PARP inhibitor (4%).ĭato-DXd was given intravenously every 3 weeks at a dose of 6 mg/kg (n=22) or 8 mg/kg (n=2), and at data cutoff 75% of patients were continuing treatment, while 25% of patients had discontinued following disease progression.Īll participants experienced at least one treatment-related adverse event (TRAE), and grade 3 and more severe TRAEs occurred in 17% of the cohort. The current TNBC cohort had 24 patients unselected for TROP2 expression who had previously been given a median of four systemic treatments, with 88% receiving at least two. Dato-DXd – an anti-TROP2 immunoglobulin G antibody combined with a topoisomerase I inhibitor and a cleavable linker – has previously shown activity in the study’s cohort of relapsed and refractory non-small-cell lung cancer patients. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
